|
Fanconi Anemia (FA) is a rare, complex autosomal or X-linked inherited genetic disorder that is characterized by aplastic anemia, cancer predisposition, congenital defects, and widespread genomic instability [1,2]. FA was first described by Guido Fanconi in 1927 when he reported three brothers exhibiting similar physical abnormalities and abnormal red blood cell counts and morphology, and since has been clinically diagnosed in over 2,000 cases with the recognition of aplastic anemia in addition to trademark physical characteristics [3]. FA is caused by a number of defects in a group of proteins that form a group of complexes that are responsible for recognizing DNA damage and signalling for its repair during interphase. There are currently sixteen known genes, termed Fanconi Anemia Complementation Groups (FANC), associated with the Fanconi Anemia pathway [2].
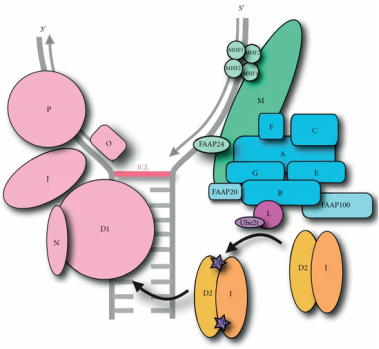 Fig 1: A model of the Fanconi Anemia complexes. Fig 1: A model of the Fanconi Anemia complexes.
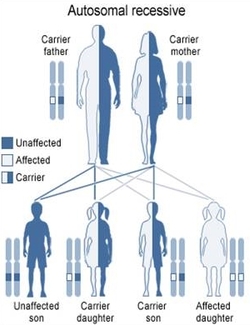
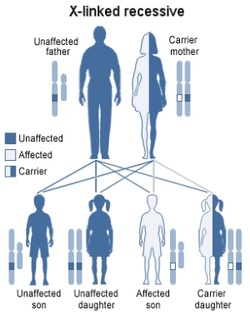
The sixteen Fanconi Anemia Complementation Groups are dispersed among eleven chromosomes, with all but one residing on autosomes. FANCB is located on the X chromosome, leading to the possibility of X-linked inheritance of the the disorder. Other than the approximately 2% of cases attributed to mutations in FANCB, FA is a recessively inherited condition at any one of the potential genes, with variable phenotypic effects [4].
The Fanconi Anemia proteins associate into three distinct complexes that each have a unique role in the repair of DNA interstrand cross-link repair; the core complex, the ID complex, and the DNA repair complex [5]. The core complex that consists of FANCA, -B, -C, -E, -F, -G, and -L is responsible for DNA damage recognition as it localizes to FANCM on interstrand cross-links (ICLs) and is vital in the activation of the entire Fanconi Anemia pathway. The ID complex, consisting of FANCI and FANCD2, acts as a messenger complex between the core complex and the DNA repair complex, requiring monoubiquitination by FANCL's e3 ubiquitin ligase ring domain to signal the location of ICLs and recruit the DNA repair complex.Lastly, the DNA crosslink repair complex is comprised of five Fanconi Anemia proteins including FANCD1, -J, -N, -O, and -P and interacts directly with the more well known BRCA1 and BRCA2 proteins to repair the damaged DNA [5]. Prevalence and Inheritance
Prognosis and ManagementIndividuals with Fanconi Anemia first and foremost experience bone marrow failure resulting in aplastic anemia, a depletion of all three types of blood cells (red blood cells, white blood cells, and platelets), hence the name. Manifestation of bone marrow failure is typically clinically recognized by age seven but can be recognized earlier when congenital defects are present. As the disease progresses two potential malignancies can occur; hematologic malignancies including acute myeloid leukemia (AML) and myelodsplastic syndrome (MDS) and nonhematologic malignancies, primarily in the form of solid tumors of the head and neck or skin. Overall, by age 40, individuals with FA have a 90% chance of severe bone marrow failure, a 10-30% chance of leukemia, and a 25-30% chance of solid tumor growth [4]. These severe conditions result in an average life expectancy of 29 years, which can be extended by early recognition and proper management [3].
Although there is no universal cure for FA due to the incredibly complex genetic nature of the disease, several treatments exist that are utilized to reduce the effects of many of the symptoms of the condition. The hematologic symptoms of FA are the easier of the two malignancies to treat for patients with FA and can be done so through two main methods; hematopoetic stem cell transplants and androgen therapy. Stem cell transplants can be used to effectively cure aplastic anemia and prevent progression of MDS and AML, however there are reports that suggest that these transplants may increase the prevalence of squamous cell cancer growth [8]. Androgen therapy may be used in cases where hematopoetic stem cell transplants are not available and have been shown to increase blood cell counts in 50% of individuals with FA, with better results seen in red blood cells than white blood cells and platelets [3,4]. Nonhematologic malignancies are a much more difficult aspect of FA to treat due to the fragile nature of DNA and the chemicals typically required to combat solid tumors. Both chemotherapy and radiation treatments for the cancers that develop as a result of FA are extremely dangerous due to the inability to repair damaged DNA that occurs when the body is exposed to these harsh conditions. Surgery is an option to remove solid tumors, but must be conducted by experienced surgeons with consultation from hematologists and oncologist with experience with the disease [4]. The Importance of FANCL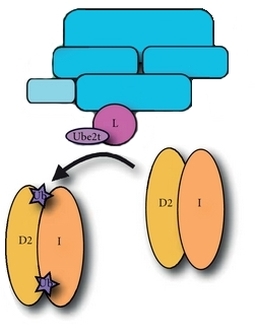 Fig 5: Ubiquitination of the ID complex by the FANCL/Ube2t complex. Fig 5: Ubiquitination of the ID complex by the FANCL/Ube2t complex.
Part of the Fanconi Anemia core complex, FANCL is a vital component of the signalling mechanism of the Fanconi Anemia pathway. FANCL interacts directly with FANCB, which together indirectly interact with FANCA to tether the proteins together within the FA core complex [9]. FANCL facilitates the ubiquitination (a posttranslational process of adding ubiquitin to a substrate) of the Fanconi Anemia ID complex that activates the complex, a requirement in the activation of the DNA repair complex. FANCL's most important domain is its zinc RING finger E3 ligase domain, a structure that recruits E2 ubiquitin-conjugating enzymes that allow ubiquitin to transfer to the ID complex. When mutations occur it can no longer facilitate this monoubiquitination event, ceasing the signalling of the core complex.
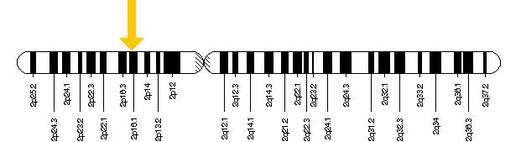
The FANCL gene is located on the short arm of the second chromosome (2p16.1) and consists of 1,753 base pairs comprising fourteen total exons. There have only been a few individuals affected by mutations to the FANCL gene, with public records indicating several unique variants including deletions, substitutions, indels, and duplications within the gene [10]. Although mutations occurring in the other core complex proteins can reduce the effectiveness of the monoubiquitination of the ID complex in vivo, FANCL has been found to be the only required core complex protein for the monoubiquitination event in vitro suggesting that it is the key protein in the activation of the Fanconi Anemia pathway [11]. |
References
[1] Jacquemont, C., & Taniguchi, T. (2007). The Fanconi anemia pathway and ubiquitin. BMC Biochemistry, 8(Suppl 1), S10. doi:10.1186/1471-2091-8-S1-S10
[2] Nalepa, G., & Clapp, D. W. (2014). Fanconi anemia and the cell cycle: new perspectives on aneuploidy. F1000Prime Reports, 6, 23. doi:10.12703/P6-23
[3] Lipton, J. M. (2013). Fanconi Anemia. In Medscape.
[4] Alter BP, Kupfer G. Fanconi Anemia. (2002). GeneReviews®.
[5] Tagrid Kaddar and Madeleine Carreau, “Fanconi Anemia Proteins and Their Interacting Partners: A Molecular Puzzle,” Anemia, vol. 2012, Article ID 425814, 11 pages, 2012. doi:10.1155/2012/425814
[6] Anneke B. Oostra, Aggie W. M. Nieuwint, Hans Joenje, and Johan P. de Winter, “Diagnosis of Fanconi Anemia: Chromosomal Breakage Analysis,” Anemia, vol. 2012, Article ID 238731, 9 pages, 2012. doi:10.1155/2012/238731
[7] Cirkovic, S., Guc-Scekic, M., Vujic, D., et al. (2011). Diagnosis of Fanconi's Anemia by Diepoxybutane Analysis in Children from Serbia. Balkan Journal of Medical Genetics, 14(2), pp. 65-70. Retrieved 16 Feb. 2015, from doi:10.2478/v10034-011-0048-6
[8] Rosenberg, P., Gerard, S., Blanche, A., Gluckman, E (2005). Risk of head and neck squamous cell cancer and death in patients with Fanconi anemia who did and did not receive transplants. Blood Jan 2005, 105 (1) 67-73; doi: 10.1182/blood-2004-04-1652
[9] Charlotte Hodson and Helen Walden, “Towards a Molecular Understanding of the Fanconi Anemia Core Complex,” Anemia, vol. 2012, Article ID 926787, 10 pages, 2012. doi:10.1155/2012/926787
[10] Auerbach, A. D. (1998). Fanconi Anemia Mutation Database. From The Rockefeller University. Retrieved February 16, 2015.
[11] Arno F. Alpi, Paul E. Pace, M. Madan Babu, Ketan J. Patel, Mechanistic Insight into Site-Restricted Monoubiquitination of FANCD2 by Ube2t, FANCL, and FANCI, Molecular Cell, Volume 32, Issue 6, 26 December 2008, Pages 767-777, ISSN 1097-2765, http://dx.doi.org/10.1016/j.molcel.2008.12.003.
[2] Nalepa, G., & Clapp, D. W. (2014). Fanconi anemia and the cell cycle: new perspectives on aneuploidy. F1000Prime Reports, 6, 23. doi:10.12703/P6-23
[3] Lipton, J. M. (2013). Fanconi Anemia. In Medscape.
[4] Alter BP, Kupfer G. Fanconi Anemia. (2002). GeneReviews®.
[5] Tagrid Kaddar and Madeleine Carreau, “Fanconi Anemia Proteins and Their Interacting Partners: A Molecular Puzzle,” Anemia, vol. 2012, Article ID 425814, 11 pages, 2012. doi:10.1155/2012/425814
[6] Anneke B. Oostra, Aggie W. M. Nieuwint, Hans Joenje, and Johan P. de Winter, “Diagnosis of Fanconi Anemia: Chromosomal Breakage Analysis,” Anemia, vol. 2012, Article ID 238731, 9 pages, 2012. doi:10.1155/2012/238731
[7] Cirkovic, S., Guc-Scekic, M., Vujic, D., et al. (2011). Diagnosis of Fanconi's Anemia by Diepoxybutane Analysis in Children from Serbia. Balkan Journal of Medical Genetics, 14(2), pp. 65-70. Retrieved 16 Feb. 2015, from doi:10.2478/v10034-011-0048-6
[8] Rosenberg, P., Gerard, S., Blanche, A., Gluckman, E (2005). Risk of head and neck squamous cell cancer and death in patients with Fanconi anemia who did and did not receive transplants. Blood Jan 2005, 105 (1) 67-73; doi: 10.1182/blood-2004-04-1652
[9] Charlotte Hodson and Helen Walden, “Towards a Molecular Understanding of the Fanconi Anemia Core Complex,” Anemia, vol. 2012, Article ID 926787, 10 pages, 2012. doi:10.1155/2012/926787
[10] Auerbach, A. D. (1998). Fanconi Anemia Mutation Database. From The Rockefeller University. Retrieved February 16, 2015.
[11] Arno F. Alpi, Paul E. Pace, M. Madan Babu, Ketan J. Patel, Mechanistic Insight into Site-Restricted Monoubiquitination of FANCD2 by Ube2t, FANCL, and FANCI, Molecular Cell, Volume 32, Issue 6, 26 December 2008, Pages 767-777, ISSN 1097-2765, http://dx.doi.org/10.1016/j.molcel.2008.12.003.
Image Resources
Header Image: "Mutation" Image
Figure 1: A model of the Fanconi Anemia complex
Figure 2: Depiction of autosomal recessive inheritance
Figure 3: Depiction of X-linked recessive inheritance [Modified image from Figure 2 for proper inheritance pattern]
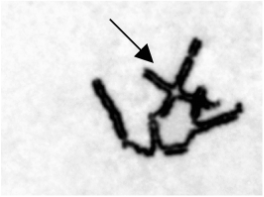
Figure 4: Image of radial chromosomes as a result of Fanconi Anemia
Figure 5: A model of the ubiquitination of the Fanconi Anemia ID complex by FANCL
Figure 6: Location of the FANCL gene
Figure 1: A model of the Fanconi Anemia complex
Figure 2: Depiction of autosomal recessive inheritance
Figure 3: Depiction of X-linked recessive inheritance [Modified image from Figure 2 for proper inheritance pattern]
Figure 4: Image of radial chromosomes as a result of Fanconi Anemia
Figure 5: A model of the ubiquitination of the Fanconi Anemia ID complex by FANCL
Figure 6: Location of the FANCL gene
|
|